Compressibility Factor Z Important Concepts and Tips for JEE Main
By A Mystery Man Writer
Last updated 03 May 2024

JEE preparation requires clarity of concepts in Compressibility Factor Z. Click here to access solved previous year questions, solved examples and important formulas based on the chapter.

What is Compressibility Factor (Z)?, Gaseous state, JEE/NEET

Real Gas and Ideal Gas, Compressibility Factor Z, States of Matter, IIT JEE, JEE MAINS
Important States Of Matter Formulas For JEE - Infinity Learn by Sri Chaitanya

Nb the value ofisothermal compressibility factor at critical point is rd (a) zero

Match the compression factor under different conditions with its va
The compressibility factors for real gases at low pressure , high pressure and that of gases of low molar masses are Z1Z2and Z3. These are[AIEEE2012 , JEEMain 2014]a)ab)bc)cd)dCorrect answer is option 'B'.
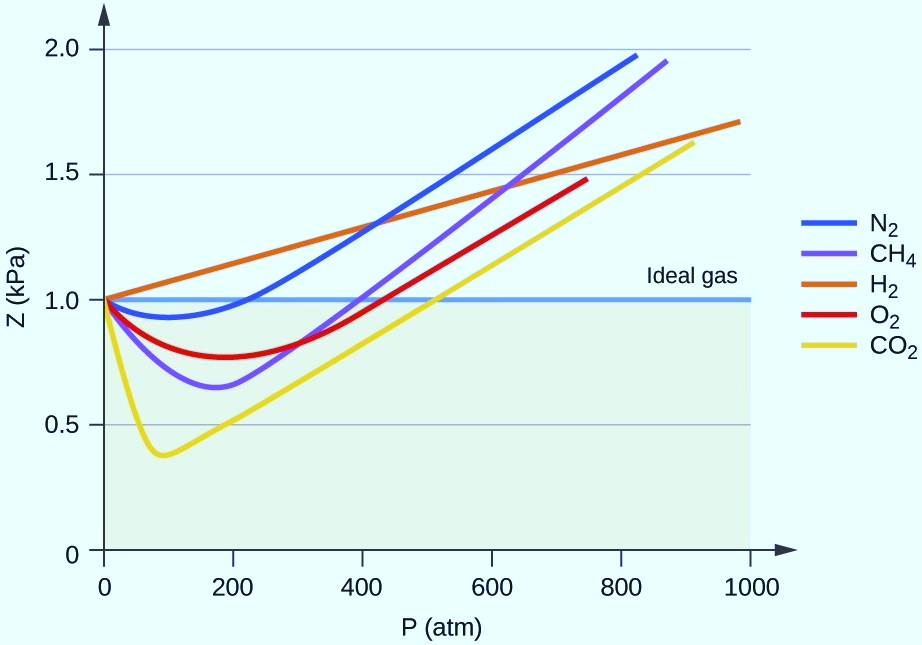
At moderate pressure, the compressibility factor for gas is given as: \[Z{\text{ }} = {\text{ }}1{\text{ }} + {\text{ }}0.35{\text{ }}P - \dfrac{{168}}{T}{\text{ }}P\], where P is in bar and T is
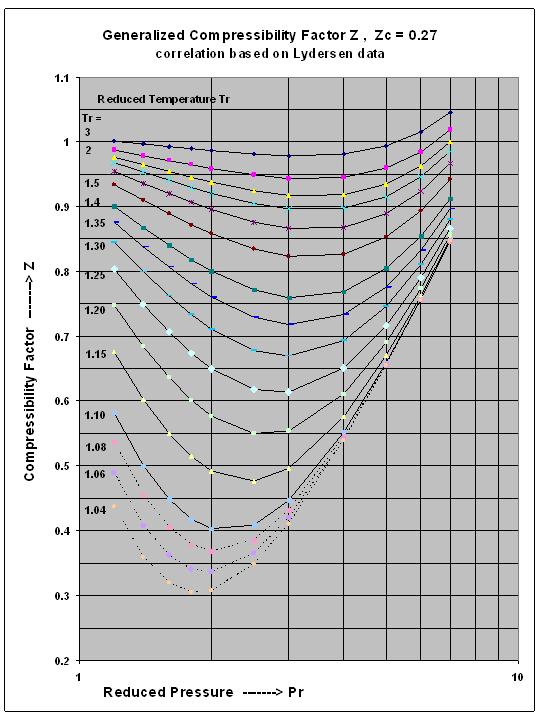
Super-critical Fluid Compressibility Factor Z , for Intermediate Reduced Pressure Range, a new correlation for excel spreadsheets

Compressibility factor (gases) - Citizendium

The compressibility factor (Z) for a gas is less than one.What does

what is the importance of compressibility factor explain with examples
Is screening effect in syllabus of JEE? - Quora
Recommended for you
 If `Z` is a compressibility factor, van der Waals' equation at low pressure can be written as14 Jul 2023
If `Z` is a compressibility factor, van der Waals' equation at low pressure can be written as14 Jul 2023 Role of Mach Number in Compressible Flows14 Jul 2023
Role of Mach Number in Compressible Flows14 Jul 2023 For H(2) gas, the compressibility factor,Z = PV //n RT is14 Jul 2023
For H(2) gas, the compressibility factor,Z = PV //n RT is14 Jul 2023 Determine Compressibility of Gases14 Jul 2023
Determine Compressibility of Gases14 Jul 2023![PDF] COMPARISON OF FIVE NATURAL GAS EQUATIONS OF STATE USED FOR FLOW AND ENERGY MEASURMENT](https://d3i71xaburhd42.cloudfront.net/62014e4d8132fefdabed288ac96e6685a25e0450/3-Table3-1.png) PDF] COMPARISON OF FIVE NATURAL GAS EQUATIONS OF STATE USED FOR FLOW AND ENERGY MEASURMENT14 Jul 2023
PDF] COMPARISON OF FIVE NATURAL GAS EQUATIONS OF STATE USED FOR FLOW AND ENERGY MEASURMENT14 Jul 2023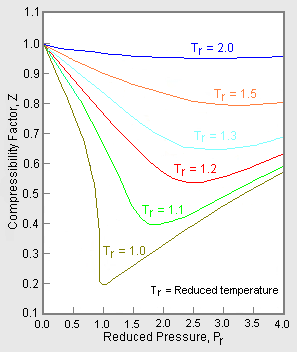 Compressibility factor (gases) - Knowino14 Jul 2023
Compressibility factor (gases) - Knowino14 Jul 2023 Compressibility - a basic concept in Fluid Mechanics14 Jul 2023
Compressibility - a basic concept in Fluid Mechanics14 Jul 2023 Compressibility factors of air using improved virial equation and14 Jul 2023
Compressibility factors of air using improved virial equation and14 Jul 2023- The compression factor (compressibility factor) for one mole of a van der Waals' gas - Sarthaks eConnect14 Jul 2023
 117. Compressibility factor H, behaving as rea gas is 1) 1 RTV 3) 1+- RT 4) (1-a) 18. If V is the observed molor unlum14 Jul 2023
117. Compressibility factor H, behaving as rea gas is 1) 1 RTV 3) 1+- RT 4) (1-a) 18. If V is the observed molor unlum14 Jul 2023
You may also like
 Lululemon scuba hoodie light blue14 Jul 2023
Lululemon scuba hoodie light blue14 Jul 2023 nu bra - Poshmark14 Jul 2023
nu bra - Poshmark14 Jul 2023 Legging Urban Girl Neon com Tule - Hipkini14 Jul 2023
Legging Urban Girl Neon com Tule - Hipkini14 Jul 2023 NFL Mens Coral Fleece Pajama Lounge Pants14 Jul 2023
NFL Mens Coral Fleece Pajama Lounge Pants14 Jul 2023 Medical Z Sujetador Postop Tirantes Ajustabl S021 Zbra Blanco 85A 1ud14 Jul 2023
Medical Z Sujetador Postop Tirantes Ajustabl S021 Zbra Blanco 85A 1ud14 Jul 2023 Upper Bounce 6 Pole Trampoline Enclosure Set to fit 16 FT14 Jul 2023
Upper Bounce 6 Pole Trampoline Enclosure Set to fit 16 FT14 Jul 2023 Girlfriend Collective Moss Tommy Bra14 Jul 2023
Girlfriend Collective Moss Tommy Bra14 Jul 2023 Exclare Women's Soft Non Padded Cup Plunge Ethiopia14 Jul 2023
Exclare Women's Soft Non Padded Cup Plunge Ethiopia14 Jul 2023 Aurora Love Tights - Rosepowder WOLFORD LONDON LTD Luxury Tights14 Jul 2023
Aurora Love Tights - Rosepowder WOLFORD LONDON LTD Luxury Tights14 Jul 2023 Ice Breaker Puffer Jacket Ice Breaker 棉服夹克398.00 超值好货14 Jul 2023
Ice Breaker Puffer Jacket Ice Breaker 棉服夹克398.00 超值好货14 Jul 2023